Abstract
Background: With BTK inhibitors and brexucabtagene autoleucel CAR-T cell therapy approved by FDA to treat B-cell malignancies, survival of patients with mantle cell lymphoma (MCL) has been greatly improved. However, frequent therapeutic relapses in patients remains as an unmet medical challenge. Preclinical tumor mouse models such as patient-derived xenografts (PDXs) are often used for drug sensitivity and treatment prediction. However, these conventional cancer models are time-consuming and expensive. In recent years, development and application of patient-derived organoid (PDO) models have led to many discoveries in various types of solid tumors. PDO models have demonstrated many advantages over PDX models. For example, PDOs can be established using biopsies as small as needle biopsies yet with much higher success rates. However, a PDO model has not been reported yet in MCL or in a few other hematologic malignancies. Therefore, in this study, with successful establishment of MCL PDO models, we optimized our protocols and applied this model for drug sensitivity screens and rapid evaluation of immunotherapies.
Methods: Patient samples were lysed with RBC lysis buffer and washed with PBS, then cell pellets were resuspended in culture medium containing cytokine cocktails. The cell suspension was aliquoted into V-bottom 96-well plates and spun. The cell aggregates within 50% Matrigel were gently transferred into multiple-well plates as needed. After solidification of the cell-Matrigel mixture, pre-warmed culture medium was added to cover them. The PDOs in 24 or 96-well plates were treated with drug or co-cultured with CAR T cells for 3-5 days. The cell viability and T cell killing activity were determined using CellTiter-Glo 3D assay kits.
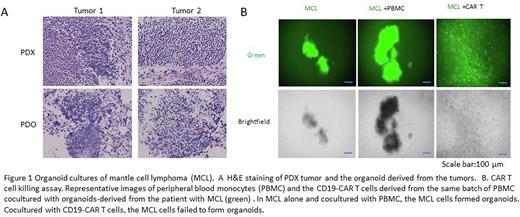
Results: We successfully established and optimized an organoid culture procedure and culture medium for diverse types of biopsy from patients with MCL. The success rate of MCL organoid development from diverse MCL biopsies was greater than 90%, much higher than for PDX. H&E staining of both tumors and their derived organoids revealed similar histological features (Figure 1A). To examine the drug sensitivity of primary MCL cells, organoid cultures derived from the patient apheresis biopsies that were either BTK inhibitor-sensitive (naive) or multiple drug resistant (to acalabrutinib, rituximab, and venetoclax) were treated with the BTK inhibitors ibrutinib or pirtobrutinib or else doxorubicin (as a control) for 48 h. Cell viability assays indicated that MCL cells in organoid cultures maintained the responses of the original tumor cells to drugs used in clinical treatment. We also demonstrated that the MCL organoids had the same major cell lineage populations as the original biopsy. These results support the notion that the PDO in vitro assay has great potential to evaluate and predict patient responses. In addition, we applied this PDO platform to evaluate CAR T cell anti-tumor activity by co-culturing the primary MCL cells with CD19-targeting CAR T cells in organoids for 3 days. To visualize and quantitate tumor killing potential, the MCL cells were labelled with the green dye 5-chloromethyl fluorescein diacetate. The cocultures were examined by imaging and FACS analysis. Compared to the same batch of peripheral blood monocytes used to generating CAR T cells, the CD19-CAR T cells very effectively eradicated CD19+ MCL cells from the co-cultures (Figure 1B), indicating that the PDO platform is a promising in vitro assay for rapid examining CAR T cell activity. Importantly, we validated drug responses in parallel in PDO and PDX models derived from the same tumors. Our preliminary results displayed high consistency between these assay systems, suggesting that PDO-based assay provides an encouraging platform for predicting clinical treatment response.
Conclusion: We have successfully established and optimized an MCL PDO platform that can be used to perform reliable and fast assays for drug sensitivity and CAR T cell anti-tumor activity in vitro.
Disclosures
Wang:Milken Institute: Consultancy; Celgene: Research Funding; Lilly: Consultancy, Research Funding; Vinverx: Research Funding; InnoCare: Consultancy, Research Funding; VelosBio: Consultancy, Research Funding; MJH Life Sciences: Honoraria; Oncology Specialty Group: Honoraria; Acerta Pharma: Honoraria, Research Funding; AbbVie: Consultancy; Molecular Templates: Research Funding; Dava Oncology: Honoraria; Eastern Virginia Medical School: Honoraria; Practice Point Communications (PPC): Honoraria; Oncternal: Consultancy, Research Funding; Genmab: Research Funding; Kite Pharma: Consultancy, Honoraria, Research Funding; Leukemia & Lymphoma Society: Consultancy, Honoraria; Merck: Honoraria; Genentech: Consultancy, Research Funding; BeiGene: Consultancy, Honoraria, Research Funding; Pharmacyclics: Consultancy, Honoraria, Research Funding; Pepromene Bio: Consultancy; Janssen: Consultancy, Honoraria, Research Funding; Deciphera: Consultancy; AstraZeneca: Consultancy, Honoraria, Research Funding; Studio ER Congressi: Honoraria; BioInvent: Consultancy, Honoraria, Research Funding; Loxo Oncology: Research Funding; IDEOlogy Health: Honoraria; LLC TS Oncology: Honoraria; Medscape: Honoraria; Meeting Minds Experts: Honoraria; Moffit Cancer Center: Honoraria; OncLive: Honoraria; Physicians Education Resources (PER): Honoraria; Juno Therapeutics: Consultancy, Research Funding.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal